EcoFAB

Microbiomes are increasingly being recognized as drivers of plant and soil health, however the complexity and large variability in these environments makes it challenging to understand how these communities function.
To facilitate and promote the development of standardized methods for studying the microbiomes, particularly in soils and plant rhizospheres, we have created fabricated ecosystems (or EcoFABs). EcoFABs (eco-fab.org) are small, single-plant scale devices that can populated with various plants, microorganisms and media (i.e., growth media, inert substrates, soils) for reductionistic science to examine the interdependencies within microbiomes and with the environment.
Protocols for The Construction of Laboratory
Ecosystems Designed to Study Plant-microbe Interactions
EcoFABs also allow for visual analyses of microbial populations (including spatial distributions and root colonization) as well as metabolomics analysis. To improve our ability to generate unique ecosystems with defined properties, we are continuing to innovate EcoFAB designs as well as develop automated platforms for high-throughput reproducible analysis of these novel ecosystems.

Laboratory Ecosystems Designed to Study Plant-microbe Interactions
Recent highlights:
EcoFABs have been shown to generate reproducible morphological and metabolomics data and the visualization of microbial colonization
Analysis of synthetic and natural microbiomes in EcoFABs can lead to better mechanistic understanding of how these communities function. Furthermore, EcoFABs can serve as a platform for communities of scientists to build on each others work to help advance microbiome science, and an improved understanding of communities can lead to healthier, productive ecosystems that can improve environmental conditions. The adoption of EcoFABs by the scientific community can lead to large-scale, interdisciplinary systems-biology studies.
ENIGMA
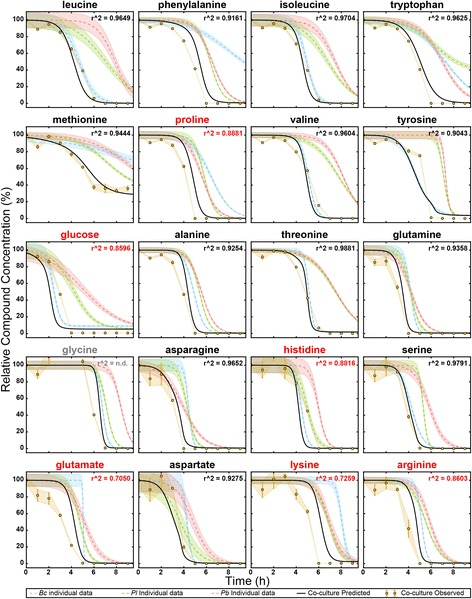
To bridge the gap between field observations and integrated field predictive models, we bring simplified communities and isolates into the lab that then provide a substrate for the more systems-level analyses of dynamical behavior.
Specifically, we aim to construct model microbial communities that allow for detailed characterization of important processes and interactions occurring in the field.
We focus on dissecting microbial interactions through the development of advanced technologies for the systematic identification and molecular dissection of inter-species interactions.
We develop and use several tools and technologies, including tractable environmentally relevant media to examine ecologically relevant patterns of substrate utilization [1], modeling of substrate utilization [2], probing microbial interactions with NIMS Mass Spectrometry Imaging (NIMS-MSI) [3] and mass spectrometry-based biomarker development for quantitatively tracking microbial populations.
